
Veeva Systems
194 Case Studies
A Veeva Systems Case Study
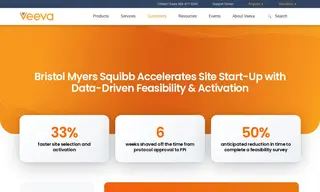
Bristol Myers Squibb (BMS) faced a challenge with fragmented feasibility data and document-heavy site start-up processes that made it hard to validate site estimates, reuse historical insights, and keep investigator sites from repeating the same work across multiple studies. The company needed a more efficient, centralized approach to feasibility and activation, and turned to Veeva Systems and its Veeva Clinical Operations, including Veeva CTMS and Veeva Study Startup.
Veeva Systems helped BMS consolidate feasibility data into a single system, automate workflows, and replace manual trackers with standardized templates and real-time dashboards. The results included a 33% faster site selection and activation process, six weeks shaved off the time from protocol approval to first patient in/first subject dosed, and an anticipated 50% reduction in time to complete feasibility surveys.

Mano Das
Director IT